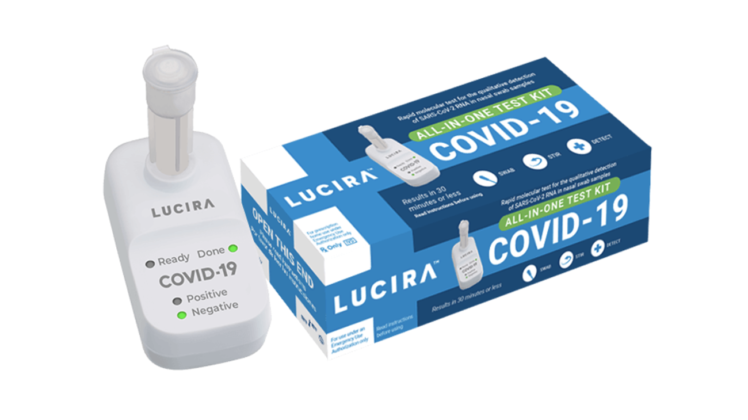
Across all data, and including 10 samples with very low viral loads (>37.5 Ct), Lucira achieved 96% accuracy, 92% PPA, and 98% NPA (3). Lucira achieved 98% accuracy, 97% positive percent agreement (PPA) and 98% negative percent agreement (NPA)(1), excluding samples with very low levels of virus that possibly no longer reflected active infection (2). In two Community Testing Studies which included individuals with and without COVID-19 symptoms, the Lucira test was compared to the Hologic Panther Fusion, one of the highest sensitivity FDA authorized SARS-CoV-2 PCR tests. Lucira’s amplification method provides a level of accuracy comparable to one of the highest sensitivity lab PCR tests. It is a molecular test that amplifies the virus’s genetic material while the test is running just like PCR lab tests. The test is intended to detect if you have an active infection and does not confirm immunity or detect antibodies. The Lucira Check It COVID-19 Test Kit is a diagnostic test intended to detect the novel coronavirus SARS-COV-2 that causes COVID-19.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
